Author's: Bassey E. Bassey
Pages: [1] - [50]
Received Date: November 16, 2017; Revised April 30, 2018
Submitted by:
DOI: http://dx.doi.org/10.18642/jmsaa_7100121890
Affirming recent positive results for the possible eradications of
dual HIV-pathogen infectivity as identified in the literature of this
work, the present paper using ordinary differential equations sought
and formulated an extended 8-dimensional mathematical dual delay
HIV-pathogen dynamic model. The study seek and addressed the
epidemiological dynamic optimal control for the application of
dual-pair treatment functions following the interplay of dual delay
HIV-pathogen infections with host target immune system cells. The
novelty of this model is informed by the combination of dual
chemotherapy and dual components of cytotoxic T-lymphocytes (CTLs) as
dual-pair treatment functions in the presence of delay intracellular
and intrinsic virulence index. We articulated the model as an optimal
control problem and therefore, adopted classical Pontryagin’s
maximum principle of the optimal control theory for its analysis.
System stability analysis was equally conducted and optimality system
of model established. Using Runge-Kutta of order 4 in a Mathcad
surface, model validity was numerically illustrated. Results
emphatically indicated tremendous maximization of healthy 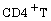 cells and maximal sustainability of precursors
and effectors of CTLs. Furthermore, elimination of both virions
infected T-cells and infectious virions were achieved at faster time
rate under minimized systemic cost and overall commercial value on
chemotherapy acquisition established. The model thus, exhibited
intellectual proceeding worthy of replication on other related
infectious diseases.
cells and maximal sustainability of precursors
and effectors of CTLs. Furthermore, elimination of both virions
infected T-cells and infectious virions were achieved at faster time
rate under minimized systemic cost and overall commercial value on
chemotherapy acquisition established. The model thus, exhibited
intellectual proceeding worthy of replication on other related
infectious diseases.
dual-pair treatment functions, HIV-pathogen infection, intrinsic virulence index, time-delay-lag, quasi-homeostatic, transversality conditions, adjoint variables.